Protein is based on nitrogen
Nitrogen is bound to make protein
Free or non-protein nitrogen is key to life.
The nitrogen story.
Nitrogen ia:
- structure - protein
- energy - more than carbohydrates, non-protein
- brain power, neuro transmitter, IQ - non-protein
- in all living matter, key to life.
Non-protein nitrogen, the power of life
- energy
- brain power
- heart energy
- bowel movements
- blood pressure control
- free to react
- swallowing
- breathing
- fertility
Composition of air
In the air we breath
According to NASA:
- Nitrogen — 78 percent
- Oxygen — 21 percent
- Argon — 0.93 percent
- Carbon dioxide — 0.04 percent
- Trace amounts of neon, helium, methane, krypton and hydrogen, as well as water vapor
Why do we have to eat nitrogen?
Nitrogen gas is not reactive for life.
- non-reactive - inert, at room temperature
- will not support life until reactive
How do we make nitrogen "reactive"?
- Oxygen is reactive, nitrogen is not, inert
- Plants bind nitrogen into non-reactive protein
- Animals eat plants to digest out the protein
- Plant eating animals have limited reactive nitrogen
- Animals convert fixed proteins into free or reactive nitrogen
- Red blood cells (red meat) is best suited to hyper-activate nitrogen into ROS
- ROS - Reactive Oxygen Species, oxidative, breathing air for faster life
- Red meat eating animals have greater availability of reactive nitrogen
- Pure Oxygen is reactive and also when fixed to carbon or nitrogen
- The most important for health is NOS - Nitric Oxide Species
Found in many forms, and including:
- Air - N2 in the air we breath
- Anesthesia - N2O in medicine
- Polution - NO2 in
- Ammonia - NH3 in the urine we pee out
- Amine - NH2− in medicine
- Nitric acid - HNO3
- Nitrates - NO−3 soluble in water
- Nitrites
The difference between carbohydrates and protein is the addition of nitrogen. Like nitrogen fertilizer is to plants, we need nitrogen to exist.
Then comes all the terms and names. Nitrogen is often in the form of NH2, this is 2 Hydrogen atoms clinging to 1 Nitrogen. Ammonia is NH3 and not healthy because it if 'hydrogenated' to become harmful to the brain and liver.
- NH2 is also called amine - healthy
- NH3 is called ammonia - un-healthy
Free-nitrogen for life
Yes, brain thinking, muscle running, even digestion requires free-nitrogen, not protein-nitrogen
Two types of nitrogen
- Protein bound not free <proteinogenic>
- Non-protein free-nitrogen <non-proteinogenic>
Non-protein /or/ non-proteinogenic amino acids
- Free nitrogen
- non-protein amino acids
- available to the immune system on a moments notice to fight germs
- Nitric Oxide (NO-)
- Reactive Oxygen Species (ROS)
Arginine has great potential to the immune systme, chains into protein, then unavailable to produce NO-
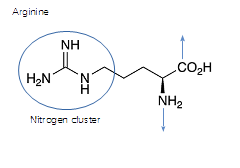 Arginine gets 'tied up' in protein chains.
Arginine gets 'tied up' in protein chains.
Creatine is the most important molecule for brain and muscle energy.
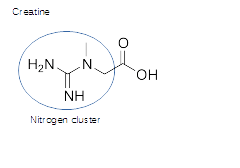 Creatine, single nitrogen cluster plus extra oxygen
Creatine, single nitrogen cluster plus extra oxygen
Urea is exclusively available to the immune system and NO- production with extra oxygen
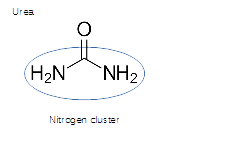 Urea, single nitrogen cluster
Urea, single nitrogen cluster
Metformin gives doulbe power to NO- where O2 easily comes from red blood cells.
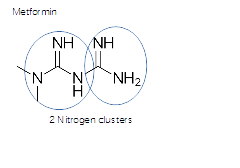 Metformin, double power from nitrogen
Metformin, double power from nitrogen
When nitrogen is low in the body we have
- auto-immune disease
- weak muscles or muscle wasting disease
- male infertility (ED)
- Low IQ
- slow healing
- needed for youths to grow
- low in old age diseases
- critically needed when sick
- low when constipated
Free nitrogen for the above cannot be stored
Must be ingested multiple times per day
Names of Nitrogen:
- Nitron- "native soda", -genes "forming" - Greek
- Nitrogen - English
- Azote - French
- Azoto - Italian
- Azot - Polish
Azoles in chemistry
- Heme as in hemoglobin, heterocyclic
- 2 atom types in a 5 member ring, heterocyclic
- imidole = azole